Graphene/ionic liquid ultracapacitors: does ionic size correlate with energy storage performance?†
Abstract
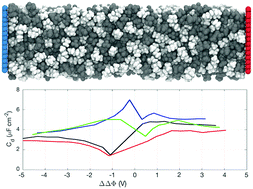
An electric double layer ultracapacitor stores energy in an electric double layer formed near its electrolyte/electrode interfaces. Graphene-based ultracapacitors, because of their outstanding performance, have attracted significant research interest. Optimization of ultracapacitor performance requires understanding the correlation of molecular characteristic of the device (such as structure and inter-ionic and ion–electrode interactions) with its macroscopic properties. Herein, we report a molecular dynamics study of how the ionic volume impacts the double-layer capacitance. Four systems were probed: large cation + large anion, large cation + small anion, small cation + large anion; small cation + small anion. Our results show that the structuring of the ionic liquid is driven by the electrolyte–electrode interactions in the ultracapacitor, which are predominantly of the van der Waals type. Storage energy densities are similar for all ultracapacitors, being in the range of 24 to 28 J cm−3 at 5.0 V. Our results present a comparative analysis of the performances of four different ILs confined between two graphene electrodes. Although the best performance has been observed for the IL with ions (cations and anions) of equal sizes, no definite conclusion about the correlation of the performance to the ionic size ratio can be made from the present study.


 Please wait while we load your content...
Please wait while we load your content...