Development and application of a routine robust graphite/poly(lactic acid) composite electrode for the fast simultaneous determination of Pb2+ and Cd2+ in jewelry by square wave anodic stripping voltammetry
Abstract
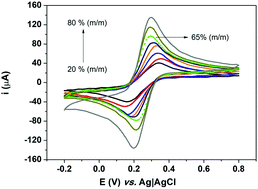
A handcrafted, low cost sustainable electrochemical sensor based on graphite/PLA was developed and applied for the simultaneous quantification of Pb2+ and Cd2+ in jewelry (metallic ores) on the basis of square-wave anodic stripping voltammetry (SWASV). The proposed electrode was characterized by different complementary strategies such as cyclic voltammetry (CV), thermogravimetric analysis (TGA), Raman spectroscopy, X-ray Micro Computed Tomography (μCT), scanning electron microscopy (SEM) and atomic force microscopy (AFM). Experimental results showed that the developed electrode exhibits excellent electrochemical performance, with accurate and well-resolved voltammetric responses for the analytes assessed. Under optimized conditions, experiments led to linear ranges from 0.9 to 35.0 μg L−1 for Cd2+, and from 1.2 to 35.0 μg L−1 for Pb2+, with detection limits estimated as 0.3 μg L−1 and 0.4 μg L−1, respectively. The fabricated electrode was successfully applied to the fast simultaneous quantification of Pb2+ and Cd2+ in jewelry samples and the findings were in agreement to those reached by graphite furnace atomic absorption spectrometry (GF AAS).


 Please wait while we load your content...
Please wait while we load your content...