Synthesis and characterization of a novel oxo-bridged binuclear iron(iii) complex: its catalytic application in the synthesis of benzoxazoles using benzyl alcohol in water†
Abstract
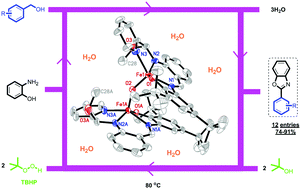
In the present work, the synthesis and characterization of a novel binuclear oxo-bridged iron(III) complex, (FeLAPIP)2O, where LAPIP is the deprotonated form of a tetradentateo-aminophenol-iminopyridine ligand, is described. The iron complex was characterized using different techniques including X-ray crystallography, infrared spectroscopy, UV-Vis, magnetic susceptibility and cyclic voltammetry. The X-ray structure analysis revealed that in the structure of the (FeLAPIP)2O complex, each iron(III) is coordinated in a distorted square pyramidal arrangement by an oxo group, three amine nitrogens and one oxygen atom of the o-aminophenolate ligand. The variable-temperature magnetic measurement exhibits strong antiferromagnetic coupling between two iron(III) centers. Cyclic voltammetry measurements showed two kinds of quasireversible events, suggesting the formation of a phenoxyl radical species in the region of the anodic peaks and a metal-centered redox (FeIII/FeII) process at low potential. The catalytic activity of this Fe-complex was evaluated in coupling of 2-aminophenol and benzyl alcohols for the one-pot synthesis of benzoxazoles. The catalyst system showed high catalytic activity in this transformation and benzoxazole derivatives were obtained in good to excellent yields. tert-Butyl hydroperoxide was used as an oxidant and 1.2 mol% of catalyst was needed to accomplish the reaction in water as the green solvent.


 Please wait while we load your content...
Please wait while we load your content...