EPR and 51V NMR studies of prospective anti-diabetic bis(3-hydroxy-4-pyridinonato)oxidovanadium(iv) complexes in aqueous solution and liposome suspensions†
Abstract
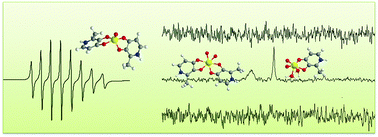
We report EPR and 51V NMR characterization of vanadium(IV/V) species originating from bis(3-hydroxy-4-pyridinonato)oxidovanadium(IV) complexes in aqueous solution at pH 7.4, under aerobic conditions and along time. Since complexes possess variable hydrophilic/lipophilic balance and consequently different solubilities in water we also performed a parallel study in POPC liposome suspensions. For less soluble complexes we used DMSO as a co-solvent. We identified three groups of complexes with different solubilities in water. Compounds 1 and 2 are soluble enough to produce 1.5 mM solutions in MOPS buffer. Compounds 3 and 4 exhibit intermediate solubility but studies in MOPS buffer and POPC liposomes demonstrate that solubilisation is considerably enhanced in liposome suspensions. Compounds 5 and 6 exhibit low water solubility, which is not sufficiently enhanced in liposome suspensions thus constraining the use of DMSO as a co-solvent. Analysis of the EPR spectra of bis(3-hydroxy-4-pyridinonato)oxidovanadium(IV) complexes shows that upon dissolution a single species is present in solution, [VOL2]. In the presence of air [VOL2] is oxidized to three species, [VO2L2]−, [VO2L] and V1 (H2VO4−) as characterized by the 51V NMR spectra. Addition of sodium ascorbate reduces the V(V) species to the original [VOL2] complex. The results demonstrate that the use of DMSO lowers the rate of oxidation of the [VOL2] complex. For DMSO percentages higher than 10% two distinct EPR signals are observable and EPR spin-Hamiltonian parameters confirm the presence of [VOL2] in two different solvation environments, which confirms the participation of DMSO in the first solvation sphere of the complex.
- This article is part of the themed collection: Equilibrium Solution Coordination Chemistry


 Please wait while we load your content...
Please wait while we load your content...