Reaction-based ratiometric fluorescent probe for selective recognition of sulfide anions with a large Stokes shift through switching on ESIPT†
Abstract
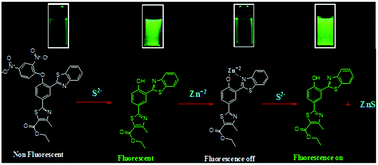
An ESIPT-based, highly selective, ratiometric fluorescent probe BNPT has been synthesized and characterized, which shows turn-on fluorescence response in the presence of S2−, attributed to the removal of a protective 2,4-dinitrobenzene (DNB) moiety, and the resulting fluorescent product has been used for the selective detection of Zn2+. UV-vis and fluorescence spectroscopy analysis, and DFT and TDDFT calculations were carried out to understand the sensing mechanism. The probe BNPT displays a rapid response time and good sensitivity. The probe can detect quantitatively in a concentration range of 0–6.0 μM. The detection limit is 31.3 nM. The sensing ability of BNPT has been successfully applied in real water samples. Applicability as an in-field test kit has been demonstrated by sensing with a BNPT-coated TLC plate. The probe BNPT has been successfully used for visualization of intracellular environment in living cells. The uniqueness lies in the fact that starting with the probe BNPT, sensing can be achieved by two different mechanisms – first, by selective de-protection of the probe BNPT and second, by reacting with the Zn2+ ensemble with S2−.


 Please wait while we load your content...
Please wait while we load your content...