Synthesis and antiviral activity of camphor-based 1,3-thiazolidin-4-one and thiazole derivatives as Orthopoxvirus-reproduction inhibitors†
Abstract
The Orthopoxvirus genus belongs to the Poxviridae family and includes variola virus (smallpox), cowpox virus, monkeypox virus and vaccinia virus (VV). Smallpox is considered one of the great epidemic disease scourges in human history. It has currently been eradicated; however, it remains a considerable threat as a biowarfare or bioterrorist weapon. The poxvirus, VV, serves as a model virus from which new antiviral therapies against Orthopoxviruses can be identified. Here, a series of nitrogen–sulphur containing heterocycles such as 1,3-thiazolidin-4-one and thiazoles containing a 1,7,7-trimethylbicyclo[2.2.1]heptan scaffold were synthesized and screened for their antiviral activity. The bioassay results showed that the 4b, 4c and 4e thiazoles, which contained a substituted benzene ring, were able to inhibit VV reproduction with IC50 values in the 2.4–3.7 micromolar range whilst exhibiting moderate cytotoxicity. Among the thiazolidin-4-one derivatives, compound 8b, which contained a 4-methylbenzylidene group, displayed good inhibitory activity (IC50 = 9.5 μM) and moderate toxicity.
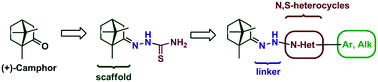


 Please wait while we load your content...
Please wait while we load your content...