DIBI, a 3-hydroxypyridin-4-one chelator iron-binding polymer with enhanced antimicrobial activity†
Abstract
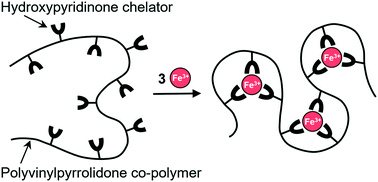
Depriving microorganisms of bioavailable iron is a promising strategy for new anti-infective agents. The new, highly water-soluble, low molecular weight co-polymer DIBI was developed to selectively bind iron(III) ions as a tris chelate and acts as a standalone anti-infective. Minimum inhibitory concentration (MIC) studies show DIBI is effective against representative reference strains for Gram-positive and Gram-negative bacteria S. aureus and A. baumannii, and the fungus C. albicans. Compared to the small molecule iron chelators, deferiprone and deferoxamine, DIBI outclassed these by factors of 100 to 1000 for inhibition of initial growth. DIBI and a series of related co-polymers (Mw of 2–9 kDa) were synthesized via reversible addition–fragmentation chain transfer (RAFT) polymerization of a chelating 3-hydroxypyridin-4-one (HPO) methacrylamide monomer and N-vinylpyrrolidone (NVP). Full incorporation of the HPO monomer into the co-polymers from the reaction solution was determined by 1H NMR spectroscopy and ranged from 4.6 to 25.6 mol%. UV-vis spectroscopy showed that all the HPO in DIBI binds readily to iron(III) in a tris chelate mode to the maximum theoretical iron(III) binding capacity of the co-polymer. Chemical characterization including single crystal X-ray diffraction analyses of the O-benzyl protected and the functional HPO monomer are discussed. By design, DIBI is highly water soluble; the highest mass fraction in water tested was 70% w/w, without the need of organic co-solvents.


 Please wait while we load your content...
Please wait while we load your content...