A Pb2+ ionic gate with enhanced stability and improved sensitivity based on a 4′-aminobenzo-18-crown-6 modified funnel-shaped nanochannel†
Abstract
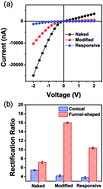
The existence of heavy ions, such as Pb2+, in the external environment is potentially hazardous as these can be highly toxic to the human body. Inspired by the highly efficient ability of biological ion channels to recognize the metal ion, much effort has been devoted to investigating biomimetic ionic gates based on engineered solid-state conical nanopores/nanochannels. However, the reported system generally displays relatively poor functionality and low stability due to the limited functional region. This article describes an ionic gate with enhanced stability and improved sensitivity based on an emerging advanced funnel-shaped nanochannel system. The ionic gate is developed by anchoring the Pb2+ ion-responsive functional molecules, 4′-aminobenzo-18-crown-6 (4-AB18C6), onto the inner surface of a funnel-shaped polyethylene terephthalate (PET) nanochannel. The system can selectively recognize Pb2+ with an ultra-low concentration of down to approximately 10−15 M and displays excellent stability. The Pb2+ ions will form positively charged complexes through specific association with 4-AB18C6, which would screen the negative charge existing on the channel walls, resulting in a decreased ionic current and also an “OFF state”. Since the ability of EDTA to associate with Pb2+ is much stronger than that of 4-AB18C6, the nanochannel can also achieve reversible switching upon the alternating addition of Pb2+ ions and EDTA. The switching behaviors of the system were reflected by the good reproducibility of the tunable rectifying effect. The stability of the conical and funnel-shaped nanochannels is also compared using current scanning under constant voltage. The results have shown that the stability of the funnel-shaped nanochannel is much better than that of the conical nanochannel, and this can be ascribed to its much longer critical region. Consequently, the funnel-shaped nanochannels with enhanced stability and improved sensitivity can potentially be applied in ion transportation, sensors, drug release, and energy conversion.
- This article is part of the themed collection: Electrochemistry at nano-interfaces


 Please wait while we load your content...
Please wait while we load your content...