Effects of operating and design parameters on ion exchange columns for nutrient recovery from urine†
Abstract
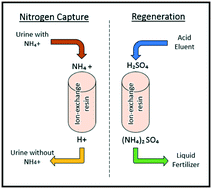
Ion exchange is a promising option for recovering nutrients (nitrogen, phosphorus, and potassium) from source-separated urine. We determined that it was feasible to integrate nitrogen and potassium recovery via cation exchange with phosphorus recovery, either via struvite precipitation or anion exchange. Flow rate and intermittent operation did not significantly affect ammonium recovery and adsorption kinetics with Dowex Mac 3, a resin with demonstrated high adsorption density and recovery efficiency. During regeneration, concentration of the sulfuric acid eluent had a more significant effect on performance metrics than did acid flow rate. Nitric acid and hydrochloric acid exhibited similar cation recovery efficiencies as sulfuric acid (>99%); sodium chloride, which has lower costs and environmental impacts associated with its production, exhibited lower recovery efficiencies (77% for ammonium, 88% for potassium). A suite of common pharmaceuticals was used to determine trace organic contaminant fate during adsorption, regeneration, and in the final fertilizer product. Of the ten pharmaceuticals measured, only atenolol and metoprolol were detected in the ammonium sulfate product, both at <0.1 μg L−1. Ultimately, macronutrients were selectively recovered from urine, which could enable customized fertilizer production.


 Please wait while we load your content...
Please wait while we load your content...