Electrochemical deposition for the separation and recovery of metals using carbon nanotube-enabled filters†
Abstract
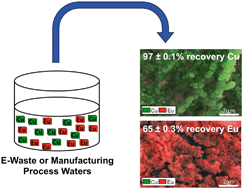
Rare earth and specialty elements (RESE) are functionally integral to several clean energy technologies, but there is no domestic source of virgin RESE in the United States. Manufacturing waste streams, which are relatively simple compositionally, and electronic wastes, which are chemically complex, could both serve as viable sources of secondary RESE if efficient methods existed to recover and separate these metals for reuse. Leveraging differences in RESE reduction potentials, high surface area, high conductivity carbon nanotubes (CNTs) could enable space- and solvent-efficient, selective recovery of RESE from mixed metal wastes. In this study, unaligned CNTs encapsulated in polyvinyl alcohol were used to develop an electrochemically active filter and tested for recovery of six metals or metalloids (Cu, As, Eu, Nd, Ga, and Sc) as a function of flow rate (1–5 mL min−1), pH (2–10), and voltage (0.1–3.0 V), with maximum recoveries of 86–96%, except for As, which was unretained. All metals were recovered as oxides, rather than their zero valent or reduced forms (except for Cu, which was partially reduced at low pHs). Deaeration experiments suggested electrochemical reduction of dissolved O2 and O2 derived from water splitting were jointly responsible for metal capture, where metal oxides were first formed via metal hydroxide intermediates, and this mechanism was enhanced at higher pHs. A synthetic, multi-metal waste stream of Cu and Eu was successfully separated on multiple stages with increasing voltages (97 ± 0.1% Cu and 65 ± 0.3% Eu recovery), indicating the approach might be useful for the treatment of electronic end-of-life and manufacturing derived wastes.


 Please wait while we load your content...
Please wait while we load your content...