Sulfidation mechanisms of Fe(iii)-(oxyhydr)oxide nanoparticles: a spectroscopic study†
Abstract
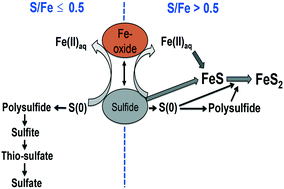
We used synchrotron-based X-ray absorption spectroscopy, transmission electron microscopy, and wet chemical analyses to study the sulfidation mechanism(s) and sulfur oxidation products from the reaction of ferrihydrite, goethite, and hematite nanoparticles with dissolved sulfide at different S/Fe molar ratios under anaerobic condition. Our results suggest that surface area alone does not explain the differences in reactivity of Fe(III)-(oxyhydr)oxide nanoparticles with dissolved sulfides; differences in atomic-level surface structure are also likely to play an important role. The higher reactivity of ferrihydrite leads to a faster sulfidation rate compared to that of goethite and hematite. We found that polysulfides as well as elemental sulfur are the major reaction products in the sulfidation of all three Fe(III)-(oxyhydr)oxide nanoparticles studied. We also found that thiosulfate and sulfate formed during the sulfidation of goethite and hematite but did not form in the case of ferrihydrite, suggesting that the slower reaction kinetics of goethite and hematite favors the formation of solid-phase thiosulfates and elemental sulfur in our experiments. In addition, our results revealed that the S/Fe ratio is a critical variable in the sulfidation reaction. Iron dissolution rates for ferrihydrite, goethite, and hematite nanoparticles were found to increase up to a S/Fe ratio of ≤0.5 and decline above this ratio, suggesting formation of FeS species. Similarly, Fe dissolution rates increased with increasing S/Fe ratios and remained an order of magnitude higher for ferrihydrite than for goethite and three times higher for ferrihydrite than for hematite. Sulfur-K-edge X-ray absorption near edge structure (XANES) spectroscopy revealed for the first time the mass distribution of these solid-phase sulfur oxidation products. In addition, we used Fe-K-edge XANES and extended X-ray absorption fine structure (EXAFS) spectroscopic analysis to follow the kinetics of FeS formation for the three types of Fe(III)-(oxyhydr)oxide nanoparticles, with varying S/Fe ratios. Ferrihydrite transformed completely to FeS in our experiments, but only 58% of the goethite and only 18% of the hematite transformed to FeS. These results have important environmental implications for Fe- and S-redox cycling and contaminant mobility and provide experimental evidence for the impact of S/Fe ratio on contaminant mobility in the systems studied, either by releasing surface-sorbed contaminants due to Fe(III)-reductive dissolution at lower S/Fe ratios or by trapping or co-precipitation of contaminants with FeS precipitation at higher S/Fe ratios.


 Please wait while we load your content...
Please wait while we load your content...
