Foliar surface free energy affects platinum nanoparticle adhesion, uptake, and translocation from leaves to roots in arugula and escarole†
Abstract
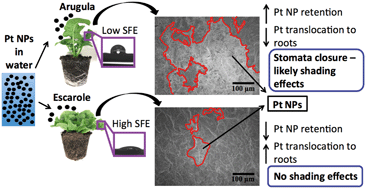
Pt nanoparticles (NPs) are directly emitted into air from human activities, contributing to the pollutant load of plants and posing risks to food safety. Foliar surface characteristics affect particle interception and retention from air; however the influence of foliar surface free energy (SFE), a quantitative measure of polar and dispersive surface forces which determine hydrophobicity/hydrophilicity, has not been investigated. Here, soil-grown plantlet leaves of arugula (low SFE) and escarole (high SFE) were exposed to Pt NPs (5–500 mg Pt NPs per L) for 5 days. Pt NP internalization and translocation, aggregation/agglomeration on leaf surfaces, and SFE changes were analyzed. Plantlets were also root-exposed to Pt NPs (one exposure to 20 mL of 50 mg L−1 dispersion) to provide a comparison between leaf-to-root and root-to-leaf translocation. Among foliar-exposed plants, inductively coupled plasma-mass spectrometry data showed that relative to escarole, arugula contained higher Pt concentrations in leaves (33, 31, and 14 times higher for 5, 50, and 500 mg L−1 exposures, respectively) and lower Pt concentrations in roots (6, 4, and 3 times lower for 5, 50, and 500 mg L−1 exposures, respectively). For both plants, the proportion of Pt translocated from roots to leaves (99% and 28% for arugula and escarole, respectively) was higher than that from leaves to roots (<1% for both plants). Scanning electron micrographs of foliar-exposed leaves (500 mg Pt NPs per L) showed a much higher degree of Pt NP aggregation/agglomeration on arugula relative to escarole, in addition to stomata closure from likely shading effects on arugula. Analysis of foliar SFE results based on acid–base theory did not indicate any relationship between Pt NP exposure and changes to SFE. We conclude that plants with low SFE are significantly more likely to adhere and retain NPs than plants with high SFE, providing implications for the application of nanopesticides, where NP adsorption and retention are of interest, and for food safety, where the presence of NPs should be minimized.


 Please wait while we load your content...
Please wait while we load your content...