Cobalt-substituted SrTi0.3Fe0.7O3−δ: a stable high-performance oxygen electrode material for intermediate-temperature solid oxide electrochemical cells†
Abstract
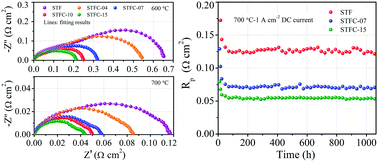
A key need in the development of solid oxide cells (SOCs) is for electrodes that promote fast oxygen reduction and oxygen evolution reactions at reduced operating temperature (≤700 °C), with sufficient durability to allow operation over desired 40 000 h lifetimes. A wide range of electrode materials have been investigated, with some providing resistance low enough for cell operation below 700 °C, but it is generally found that the electrode performance degrades over time. Here we demonstrate an oxygen electrode material, Sr(Ti0.3Fe0.7−xCox)O3−δ (STFC), that provides a unique combination of excellent oxygen electrode performance and long-term stability. The addition of a relatively small amount of Co to Sr(Ti0.3Fe0.7)O3−δ, e.g., x = 0.07, reduces the electrode polarization resistance by >2 times. The STFC electrode yields stable performance in both fuel cell and electrolysis modes at 1 A cm−2. The fundamental oxygen diffusion and surface exchange coefficients of STFC are determined, and shown to be substantially better than those of La0.6Sr0.4Co0.2Fe0.8O3−δ, the most widely used SOC oxygen electrode material. While other electrode materials have been shown to exhibit better oxygen transport coefficients than STFC, they do not match its stability.


 Please wait while we load your content...
Please wait while we load your content...