Enhanced electrocatalytic performance for the hydrogen evolution reaction through surface enrichment of platinum nanoclusters alloying with ruthenium in situ embedded in carbon†
Abstract
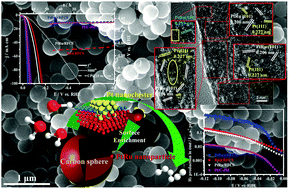
The hydrogen evolution reaction is a crucial step in electrochemical water splitting. The most efficient catalysts for this reaction in acidic media are Pt based, but the high cost of Pt limits its practical applications. We developed a novel matrix architecture in which a trace amount of Pt is alloyed in situ with Ru nanoparticles uniformly and partially embedded in porous carbon spheres. The synthetic procedure is simple and efficient. Surface enrichment of metallic Pt nanoclusters on PtRu alloy nanoparticles results in weak bonding with hydrogen and rapid hydrated proton dissociation. These effects significantly increase the electrocatalytic activity in the hydrogen evolution process. This robust catalyst, with a Pt loading 99.9% less than that of a commercial Pt-based catalyst, gave a high turnover frequency (4.03 H2 s−1), a small Tafel slope (27.2 mV dec−1), and comparable overpotentials (19.7 mV and 43.1 mV), as well as achieved current densities of 10 and 100 mA cm−2, i.e., better than those obtained with commercial Pt/C catalyst, in 0.5 M H2SO4. This structure prevents nanoparticles from dissolving, agglomerating, and detaching during long-term operation; therefore there was no obvious decrease in catalytic activity after continuous reaction.


 Please wait while we load your content...
Please wait while we load your content...