Spatial control of cocatalysts and elimination of interfacial defects towards efficient and robust CIGS photocathodes for solar water splitting†
Abstract
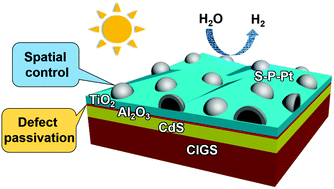
Chalcopyrite thin film absorbers such as Cu(In,Ga)Se2 (CIGS) exhibit excellent solar energy conversion efficiency, particularly when coupled with CdS to form an excellent p–n junction. To advance CIGS towards an efficient photoelectrochemical (PEC) hydrogen evolution reaction (HER), a protective overlayer (typically TiO2) is needed to prevent the corrosion of CIGS and CdS in the electrolyte, and a HER catalyst (typically Pt) is required to promote the surface reaction. However, it is a great challenge to realize delicate spatial control of the HER catalyst on the surface of the protective overlayer using the traditional deposition method. The charge transport through the CdS/overlayer interface is also of vital importance but is rarely considered. Herein, through a new two-step platinization strategy, the dispersion and particle size of Pt nanoparticles are independently controlled to realize high HER catalytic activity. Moreover, defects at the CdS/TiO2 interface are passivated via an ultrathin Al2O3 insertion layer. Consequently, we have obtained a robust CIGS/CdS/Al2O3/TiO2/Pt photocathode for PEC hydrogen evolution, which yields an applied bias photon-to-current efficiency (ABPE) of 6.6% in neutral electrolyte with a long-term stability up to 8 h (4.5% drop), and an unprecedented ABPE of 9.3% in acidic electrolyte that is the highest among chalcopyrite-based photocathodes. When paired with a BiVO4 photoanode to form a PEC tandem cell, an unbiased solar-to-hydrogen conversion efficiency of 1.01% is achieved.


 Please wait while we load your content...
Please wait while we load your content...