Direct photoactivation of a nickel-based, water-reduction photocathode by a highly conjugated supramolecular chromophore†
Abstract
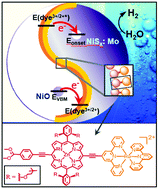
In dye-sensitized photoelectrosynthesis cells, multi-step photoinduced electron transfer takes place to generate redox-separated (RS) states that activate catalysts for water splitting or carbon dioxide reduction. From photoexcitation of the chromophores to formation of the RS states, the solar energy initially stored at the chromophore excited states is reduced stepwise in a sequence of photoinduced electron transfer steps. We report here a water-reduction photocathode based on a supramolecular chromophore, an ethyne-bridged (porphyrinato)zincII and bis(terpyridyl)rutheniumII complex, which is surface-bound to a mesoporous nickel oxide electrode, with an over-layer of nickel sulfide derivative as a water reduction catalyst. Visible light excitation of the chromophore generates a long-lived RS state that forms directly at its excited state with the electron delocalized at the terpyridyl ligands for transferring to the nickel sulfide catalyst, and the hole at the zinc porphyrin moiety for injecting into the nickel oxide electrode. The resulting photocathode shows enhanced photoelectrocatalytic performances relative to the previously reported NiO-based photocathodes. A key element lies in the efficient, direct activation of the catalyst by the long-lived, RS excited state that minimizes the energy loss along the photoinduced electron transfer steps towards water reduction.


 Please wait while we load your content...
Please wait while we load your content...