Tuning the properties of poly(2,6-dimethyl-1,4-phenylene oxide) anion exchange membranes and their performance in H2/O2 fuel cells†
Abstract
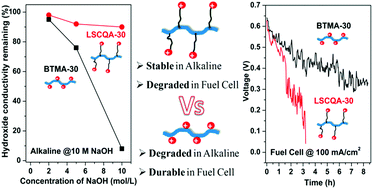
Here, we present a complete investigation of poly(2,6-dimethyl-1,4-phenylene) (PPO) AEMs with quaternary ammonium (QA) groups placed at different locations within the chemical structure of the polymer, e.g. a standard benzyltrimethyl ammonium control (BTMA), side-chain-type (SCQA), long side-chain-type (LSCQA), comb-shaped (CQA), or side-chain type/comb-shaped (SCCQA) AEMs have been designed and studied. These copolymers possess a similar composition but considerably different molecular architectures, the nature of which significantly alters their properties and device performance. Hydroxide conductivity was improved for the SCQA, LSCQA and comb-shaped membranes possessing a C-18 alkyl terminal pendant compared to that of BTMA and comb-shaped samples with a short alkyl chain. In chemical stability experiments under 10 M NaOH and 80 °C for 200 hours, LSCQA and SCCQA samples with a C-18 alkyl terminal pendant architecture showed less decrease in conductivity (∼10%) than the BTMA, SCQA and SCCQA polymers having short alkyl chains, which lost more than 50% conductivity after alkaline stability testing. Significant degradation was observed for the unstable PPO AEM samples by either SN2 substitution or Hoffmann elimination according to 1H NMR analysis. Interestingly, the fuel cell device performance provided counterintuitive data that showed that longer side chains with excellent alkaline stability were not superior in device function assessment. Specifically, the highly alkaline stable long side-chain-type LSCQA-30 membrane showed significant degradation in a fuel cell device with an operating lifetime of 3.3 h at 100 mA cm−2. Analysis of the aged membrane showed SN2 substitution and elimination of trimethylamine to be the dominant degradation mechanisms. In contrast, the BTMA-30 membranes with poor alkaline stability showed good durability in a working device without obvious degradation after 8.3 h operation as confirmed by 1H NMR spectra. This new finding that contrasts membrane alkaline stability and device operation stability is extremely important and gives us directions for new polymer designs for high performance devices. Additionally, this study paves the way for coupled ex situ chemical stability and in-device degradation studies, which are sorely needed in this field.


 Please wait while we load your content...
Please wait while we load your content...