A specific demetalation of Fe–N4 catalytic sites in the micropores of NC_Ar + NH3 is at the origin of the initial activity loss of the highly active Fe/N/C catalyst used for the reduction of oxygen in PEM fuel cells†
Abstract
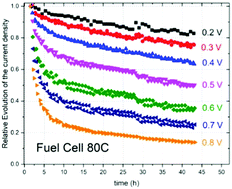
In this study, we explored the behavior of NC_Ar + NH3, an initially highly active catalyst for oxygen electroreduction, in H2/air fuel cells from 0.8 to 0.2 V at 80 °C and 25 °C, in order to find the causes of its instability. We discovered that the decay of the current density always involves the superposition of fast and slow first order kinetics, for which half-lives were obtained. The half-life of the fast decay was practically the same at all potentials and temperatures with a value of around 138 ± 55 min, while the half-life of the slow decay greatly varied from a minimum of ≈2400 min (40 h) to infinity. From the adsorption–desorption isotherm of NC_Ar + NH3, it was deduced that the Fe/N/C carbonaceous catalyst is characterized by interconnected open-end slit-shaped micropores, in which water (with dissolved H+ and O2) quickly flows in the fuel cells if their width is ≥0.7 nm as it has no interaction with the hydrophobic walls of the micropores. The driving force of this quick water flow is the humidified air streaming through the working cathode. Fe–N4-like active sites are thermodynamically stable in stagnant acidic conditions, but according to the Le Chatelier principle, they demetalate in the flux of water running into the micropores. This specific demetalation is the cause of the initial loss of ORR activity of NC_Ar + NH3 catalysts assigned to the fast current decay in fuel cells.


 Please wait while we load your content...
Please wait while we load your content...