Photoinduced electron transfer and remarkable enhancement of magnetic susceptibility in bridging pyrazine complexes†‡
Abstract
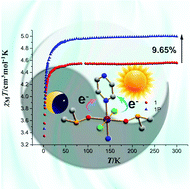
Three isostructural coordination polymers of the formula [MCl2(DMSO)2(pz)]n (pz = pyrazine, DMSO = dimethyl sulfoxide, M = Mn, Fe and Co for 1–3) were synthesized through the solvothermal reaction of MCl2, DMSO and pz. By introducing DMSO and chloride as electron donor ligands and pz as an accepter, a donor–metal–accepter (D–M–A) system was constructed. Radicals generated by photoinduced electron transfer (PET) from a donor to an accepter result in a dramatic change of vibronic absorption spectra in UV regions and remarkable enhancement of magnetic susceptibility. The mechanism of PET was also determined by PXRD, IR, EPR and XPS measurements. This is the first example of bridging pz complexes which exhibit both photochromism and photomagnetism in a PET process.


 Please wait while we load your content...
Please wait while we load your content...