Synthesis and characterization of a series of transition metal oxychlorides: MBi(SeO3)2(H2O)Cl (M = Co, Ni, Cu)†
Abstract
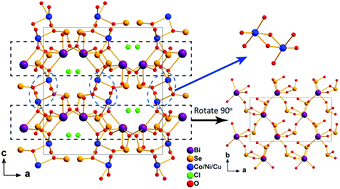
Three new transition metal oxychlorides MBi(SeO3)2(H2O)Cl (M = Co, Ni, Cu) have been firstly synthesized through a hydrothermal reaction method at 200 °C. They were structurally determined to be isostructural with the Pbca space group of the orthorhombic system. They feature a 3D framework topology with two-dimensional tunnels intersected and filled with Cl anions parallel to the crystallographic bc-plane. Two neighboring MO5Cl octahedra are connected by a sharing edge into a M2O8Cl2 dimer which serves as the structural knots to knit the 2D [Bi(SeO3)2]∞ layers in the ab-plane together into the total 3D crystal architecture. The optical band gaps of CoBi(SeO3)2(H2O)Cl (1), NiBi(SeO3)2(H2O)Cl (2) and CuBi(SeO3)2(H2O)Cl (3) were evaluated to be 3.7 eV, 3.5 eV and 3.2 eV, respectively, through extrapolating the UV-vis-NIR optical absorption spectra. Besides, the spin-allowed d–d transition absorption spectra of the transition ion centers are observed in three compounds exhibiting different colors. Compounds 1, 2 and 3 can stay thermally stable below 370 °C, 400 °C and 300 °C, respectively, as found from the results of thermal analysis based on the simultaneous thermogravimetry and differential scanning calorimetry techniques. 1, 2 and 3 exhibit antiferromagnetic properties below Néel temperatures 6 K, 18 K and 52 K, respectively. Above the Néel temperatures, Curie–Weiss behavior dominates in the M–T process for the three compounds. The cell parameters are listed: a = 14.056 Å, b = 7.582 Å, c = 14.996 Å, and Z = 8 for 1, a = 14.083 Å, b = 7.575 Å, c = 14.860 Å, and Z = 8 for 2, and a = 14.576 Å, b = 7.371 Å, c = 14.656 Å, and Z = 8 for 3.


 Please wait while we load your content...
Please wait while we load your content...