Redox-driven porphyrin based systems for new luminescent molecular switches†
Abstract
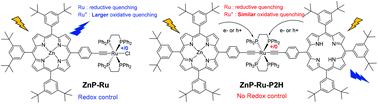
In this work, we explore the possibility of tuning the fluorescence intensity of two porphyrin systems through the electrochemical oxidation of an appended ruthenium acetylide bridge. Two electrochemically switchable systems, a dyad (ZnP-Ru, 3) and a triad (ZnP-Ru-P2H, 5), were prepared and investigated. In the ZnP-Ru dyad, the fluorescence of the zinc porphyrin was switched reversibly between the ON and OFF state upon the oxidation of the ruthenium unit, the most probable quenching process involved after oxidation being the electron transfer from the singlet excited state of ZnP to the oxidized ruthenium center. In the ZnP-Ru-P2H triad, we show that both porphyrins’ fluorescence are highly quenched independent of the redox state of the ruthenium bridge owing to the efficient photoinduced charge transfer within the ruthenium complex.
- This article is part of the themed collection: Spotlight Collection: Inorganic Molecular Electronics


 Please wait while we load your content...
Please wait while we load your content...