Investigating low-valent compositions in the Na3V2O2x(PO4)2F3−2x family: structural transitions and their consequences†
Abstract
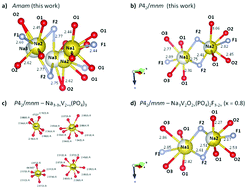
A member of the family of compounds with the formula Na3V2O2x(PO4)2F3−2x is synthesized by carbothermal reduction and 2 consecutive hydrothermal processes. The initial structural and spectroscopic characterization indicates that there are two phases in the as-synthesized material, a mixed valent phase with an intermediate V oxidation state adopting a P42/mnm space group at about 66%, and another phase with a V oxidation state close to V3+ adopting an Amam space group at about 33%. The role of each species in the electrode function is interrogated using in situ synchrotron X-ray diffraction and these data indicate that soon after charge begins, the Amam phase transforms into the P42/mnm phase. Further structural evolution of the material shows a prevailing two-phase reaction at lower potentials and a significant solid solution region during the high potential feature in the electrochemical curve with a final two-phase region at the end of charge. The conversion of the Amam component into P42/mnm showed no reversibility upon discharge. This phase-mixed electrode appears to illustrate that the mixed valent P42/mnm phase dominates electrochemical behavior during cycling and this may prove vital when preparing members in the sodium vanadium fluorophosphate family, especially if there is an energetically preferred phase for cycling.


 Please wait while we load your content...
Please wait while we load your content...