Hyper-cross-linked polymer supported rhodium: an effective catalyst for hydrogen evolution from ammonia borane†
Abstract
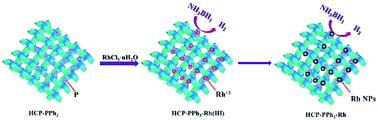
Metal nanoparticles (NPs) have wide applications in hydrogen evolution from ammonia-borane (AB) hydrolysis because they can provide large surface active areas for reactants, and thus produce high catalytic activity. Here, a hyper-cross-linked polymer (HCP-PPh3), which was synthesized through the Friedel–Crafts reaction of benzene and triphenylphosphine, was employed as a support to stabilize Rh NPs. The characterization results revealed that the Rh NPs were uniformly dispersed on the surface of HCP-PPh3, and that they had an average particle size of 2.1 nm. The as-prepared HCP-PPh3-Rh was used as an active catalyst for hydrogen generation from AB hydrolysis. This catalyst exhibited a high turnover frequency of 481 mol H2 (molRh min)−1 for AB hydrolysis under mild conditions. The high catalytic performance of HCP-PPh3-Rh can be attributed to the small size of Rh NPs and the strong interaction between the metal and HCP-PPh3. This work highlights a potentially powerful strategy for preparing highly active HCP stabilized metal NPs for AB hydrolysis to generate hydrogen.


 Please wait while we load your content...
Please wait while we load your content...