Surprisingly high sensitivity of copper nanoparticles toward coordinating ligands: consequences for the hydride reduction of benzaldehyde†
Abstract
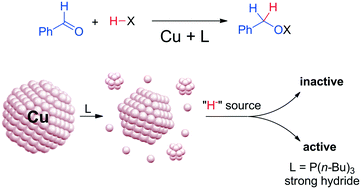
Functionalized copper nanoparticles are widely used as catalysts; however, identification of the catalytically active species remains a challenge. In this study we investigate hydride-assisted reduction reactions with special focus on the structural evolution of copper nanoparticles in the presence of phosphine and nitrogen-based ligands. These compounds are commonly used as stabilizing agents, and therefore are important in the context of other catalytic reactions as well. In particular, we investigate the case of tri-n-butylphosphine. Our findings show that ultrasmall nano-objects are formed as key intermediates to produce catalytically active species in the hydrosilylation of benzaldehyde into the corresponding silyl alcohol. Moreover, we found that the strength of the hydride donor is essential for the formation of the active catalysts. Therefore, this work unveils the previously overlooked high sensitivity of copper nanoparticles toward coordinating ligands in the context of catalysis.


 Please wait while we load your content...
Please wait while we load your content...