Acidic effect of porous alumina as supports for Pt nanoparticle catalysts in n-hexane reforming†
Abstract
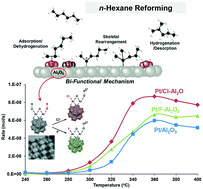
Acidic halogen-modified (Cl and F) porous alumina supports with well-defined macropores and mesopores were designed to prepare alumina-supported Pt nanoparticle (NP) catalysts (Pt/Cl–Al2O3, Pt/F–Al2O3, and Pt/Al2O3). The catalysts were then used for n-hexane reforming in a tubular fixed bed reactor with a hexane : H2 ratio of 1 : 4.3 at ambient pressure and various temperatures (240–400 °C). Although the reaction rates for all catalysts were maximised at 360 °C, Pt/Cl–Al2O3 exhibited the highest rate (at 8.66 × 10−8 mol s−1). Regarding product selectivity, Pt/Cl–Al2O3 and Pt/F–Al2O3 yielded a higher number of olefin products and fewer cracking products than Pt/Al2O3. Temperature programmed desorption (TPD) with ethanol and in situ diffuse-reflectance infrared Fourier transform spectroscopy (DRIFTS) with CO and pyridine adsorption were used to characterise the alumina support surface acidity. Compared with γ-Al2O3, the TPD results indicated that the Cl–Al2O3 and F–Al2O3 surface acidities were enhanced by surface modification. The in situ DRIFTS experiments confirmed that the relative ratio of Lewis to Brønsted acid sites of Cl–Al2O3 (0.64) was higher than those of F–Al2O3 (0.54) and unmodified Al2O3 (0.56). Additionally, the DRIFTS spectra confirmed that the Pt NPs were preferentially deposited onto the Lewis acid sites of the supports, and the CO adsorption spectra revealed that Pt NPs with (111) facets were preferentially deposited onto the Lewis acid sites. The surface acidity studies indicated that the enhanced Lewis acidity of Cl–Al2O3 induced high reaction rates at all temperatures, resulting in skeletal rearrangements of hydrocarbons toward branched isomers at high temperature via a conventional bifunctional mechanism.


 Please wait while we load your content...
Please wait while we load your content...
