Microwave-induced synthesis of a bimetallic charge-transfer metal organic framework: a promising host for the chemical fixation of CO2†
Abstract
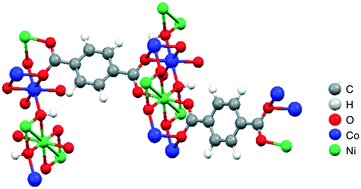
The aqueous synthesis of a bimetallic metal organic framework (MOF) with Ni and Co as the active metal centers and benzene-1,4-dicarboxylic acid as the linker has been achieved rapidly in high yield using microwave irradiation. The synthesized MOF is investigated for its catalytic efficacy in the synthesis of cyclic carbonates from epoxides and CO2. The Ni–Co-MOF provides high conversion rates of epoxides to cyclic carbonates with >99% selectivity under solvent-free conditions. The bimetallic framework (Ni–Co-MOF) exhibits superior catalytic activity to those of the corresponding single metal MOF catalysts (Ni-MOF and Co-MOF) and their mechanical combination (Ni-MOF + Co-MOF), which indicates the existence of a synergistic catalytic effect based on charge transfer between the Ni and Co metal centers and is highly advantageous for the catalytic chemical fixation of CO2. The catalytic potential of the Ni–Co-MOF is also applied to terminal and cyclic epoxides, and a recyclability study over a minimum of six cycles is also conducted. Finally, a plausible reaction mechanism for Ni–Co-MOF-catalyzed epoxide-CO2 cycloaddition reactions is also proposed.


 Please wait while we load your content...
Please wait while we load your content...