Copper promoter effect on acid–base and redox sites of Fe/Al2O3 catalysts and their role in ethanol–acetone mixture conversion
Abstract
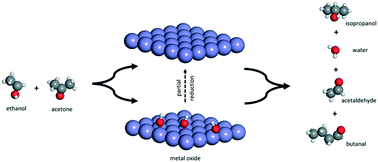
Active species of copper and iron oxide (Cu–Fe) catalysts supported on alumina were prepared by combining Pechini and wet impregnation methods. The effect of combined acid–base and redox sites of Cu and Fe species on gas-phase ethanol–acetone mixture conversion was investigated. The catalysts were characterized by chemical analyses, XRD, H2-TPR, Mössbauer spectroscopy, N2 physisorption, CO2-TPD, SEM-EDS, TG/DTA and pyridine adsorption isotherms. N2 adsorption/desorption isotherms and SEM-EDS analysis showed that the addition of copper caused an increase of BET surface area and Cu and Fe oxide dispersion. H2-TPR characterization showed that interactions between Cu and Fe oxides shift the reducibility of Fe3+ species to lower temperature improving the redox properties of the catalyst. The partial reduction of the Cu and Fe oxide species was found to be efficient in inhibiting the side decomposition reactions, improving the catalytic efficiency towards dehydrogenation and hydrogen transfer processes. It was found that acid–base pairs play an important role in the formation of dehydrogenation, dehydration and condensation products from ethanol, while redox sites are decisive for hydrogen transfer reactions with reduction of acetone to isopropanol. H2-TPR and Mössbauer spectroscopy results for the spent catalysts revealed that the highest catalytic performance of the Cu–FeAl catalysts may be attributed to the good dispersion of the Cu oxide and the site generated by the partial reduction which produces Cu+/Cu0 and Fe2+ active species. A reaction pathway with the participation of the acid–base and redox sites in the formation of products by consecutive dehydrogenation–condensation or dehydrogenation–hydrogenation reactions has been proposed.


 Please wait while we load your content...
Please wait while we load your content...