Iron oxidation dynamics vs. temperature of synthetic potassic-ferro-richterite: a XANES investigation†
Abstract
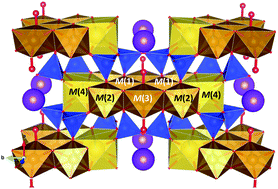
We investigated the oxidation behaviour of a synthetic potassic-ferro-richterite up to 750 °C by using simultaneous X-ray absorption spectroscopy and X-ray diffraction experiments with synchrotron radiation. From the X-ray diffraction results, we observed an abrupt decrease of cell dimensions at ∼335 °C accompanied by an anomalous increase in the monoclinic cell angle β. From the analysis of the XANES spectra at the iron K-edge, we observed that the structural shrinkage is due to the iron oxidation process, coupled to hydrogen loss, occurring at ∼315 °C, slightly before the cell contraction. Combining these results with previous studies performed on similar samples by single-crystal structure refinement, Mössbauer, high temperature-Fourier transform IR and Raman spectroscopies, we show that the temperature evolution in Fe-amphiboles is a multi-step process. Following the iron oxidation driven by temperature, the structural dynamics in this double-chain silicate is ruled by local strains in the ribbon of iron-filled octahedra, mainly due to the contraction of the M(1) site.


 Please wait while we load your content...
Please wait while we load your content...