Uptake of water by an acid–base nanoparticle: theoretical and experimental studies of the methanesulfonic acid–methylamine system†
Abstract
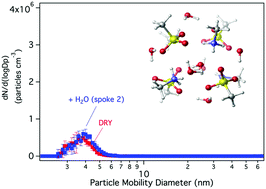
The effect of water on the growth of dry nano-size acid–base particles is not yet known. In this paper, we investigate the uptake of water by nano-size particles composed of methanesulfonic acid (MSA) and methylamine (MA) using a combination of quantum chemical calculations and laboratory experiments. Calculations were performed on the (MSA–MA)4 cluster as the dry nanoparticle model, which forms a pseudo-cubic structure, to which twelve water molecules were added successively. Theoretical results show that the hydrated clusters (MSA–MA)4–(H2O)n, n = 1 to 12 are thermodynamically stable. In ab initio dynamic simulations, no loss of water or significant changes of structure are seen for at least 10 picoseconds. In all the clusters studied, most of the water molecules lie on the face of the (MSA–MA)4 initial dry unit, and water is found to be incorporated inside the initial unit for n ranging from five to twelve. Sizes of hydrated clusters exceed significantly that of the dry cluster only for n ≥ 6. These theoretical results suggest that dry MSA–MA clusters cannot dissociate in small quantities of water. Calculations of hydrated cluster distributions at steady state show that the cluster compositions studied, with up to 12 water molecules, encompass all the hydrated clusters under the experimental conditions (RH ∼ 19%, 300 K). Experiments performed in a glass flow reactor showed no changes in size or number concentration when particles formed from MSA–MA were subsequently exposed to water vapor, in contrast to increases in both size and number when water was present during particle formation. Thus, the results seem to imply for both experiment and theory that growth in size of a particle due to uptake of water requires the previous presence of some level of hydration. These results illustrate the importance for atmospheric models of understanding on a molecular basis the mechanisms of particle formation in air.


 Please wait while we load your content...
Please wait while we load your content...