Theoretical exploitation of acceptors based on benzobis(thiadiazole) and derivatives for organic NIR-II fluorophores†
Abstract
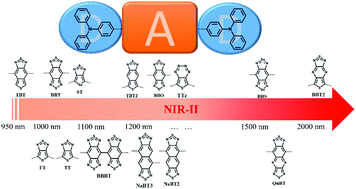
Small-molecule dyes with fluorescence emission in the second near-infrared (NIR-II) region (1000–1700 nm) have attracted considerable attention in the biomedical and bioimaging fields due to their greater imaging depths, better spatial resolution, and higher signal-to-background ratios. However, currently reported organic NIR-II fluorophores are still limited and there is great demand to develop other novel NIR-II fluorophores besides benzobisthiadiazole (BBT)-based fluorophores. More importantly, there is a lack of an appropriate level of theory capable of providing both efficient and accurate predictions of the electronic structures of organic NIR-II fluorophores. In this work, successful application of time-dependent density functional theory (TDDFT) using optimally-tuned range-separated functionals for calculations of both absorption and fluorescence spectral properties has been demonstrated, compared with the available experimental data. A series of thiadiazole-based acceptors (A) and derivatives based on the D–A–D skeleton are designed coupled with the triphenylamine donor (D). The structure–property relationships for these fluorophores are thus revealed by analyzing their ground (S0) and excited (S1) state geometries, frontier molecular orbitals (HOMO and LUMO), HOMO–LUMO energy gaps, oscillator strengths, hole–electron distributions and fluorescence wavelengths. It is suggested that the existence of a hypervalent structure leading to a much lower LUMO level and accompanying significant hole–electron separation plays a key role in the red-shift of fluorescence emission in the NIR-II region. In addition, the substitution of BBT oligomers and analogues as acceptor cores is an efficient way to achieve both red-shifted fluorescence wavelengths and enhanced oscillator strengths. The present work provides a reliable and efficient theoretical tool for predicting the related electronic and spectral properties of organic fluorophores and future screening out of potential candidates for excellent NIR-II molecular fluorophores.


 Please wait while we load your content...
Please wait while we load your content...