Chemical kinetics in an atmospheric pressure helium plasma containing humidity†
Abstract
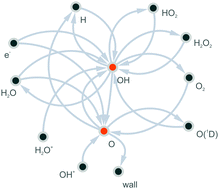
Atmospheric pressure plasmas are sources of biologically active oxygen and nitrogen species, which makes them potentially suitable for the use as biomedical devices. Here, experiments and simulations are combined to investigate the formation of the key reactive oxygen species, atomic oxygen (O) and hydroxyl radicals (OH), in a radio-frequency driven atmospheric pressure plasma jet operated in humidified helium. Vacuum ultra-violet high-resolution Fourier-transform absorption spectroscopy and ultra-violet broad-band absorption spectroscopy are used to measure absolute densities of O and OH. These densities increase with increasing H2O content in the feed gas, and approach saturation values at higher admixtures on the order of 3 × 1014 cm−3 for OH and 3 × 1013 cm−3 for O. Experimental results are used to benchmark densities obtained from zero-dimensional plasma chemical kinetics simulations, which reveal the dominant formation pathways. At low humidity content, O is formed from OH+ by proton transfer to H2O, which also initiates the formation of large cluster ions. At higher humidity content, O is created by reactions between OH radicals, and lost by recombination with OH. OH is produced mainly from H2O+ by proton transfer to H2O and by electron impact dissociation of H2O. It is lost by reactions with other OH molecules to form either H2O + O or H2O2. Formation pathways change as a function of humidity content and position in the plasma channel. The understanding of the chemical kinetics of O and OH gained in this work will help in the development of plasma tailoring strategies to optimise their densities in applications.


 Please wait while we load your content...
Please wait while we load your content...