Combined effect of hydrogen bonding interactions and freezing of rotameric equilibrium on the enhancement of photostability†
Abstract
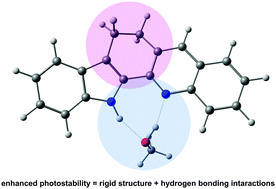
The photophysics and photostability of 12,13-dihydro-5H-indolo[3,2-c]acridine (IA), a rigid bifunctional indole derivative with proton donor/acceptor functionalities, can be drastically changed by the environment. The formation of hydrogen bonds with alcohols leads to a significant decrease of the triplet formation efficiency and an increase of photostability. The photodegradation yield was found to be about two hundred times lower in methanol and 1-propanol than in n-hexane or acetonitrile. A similar effect has been reported for two indole-naphthyridines, molecules that can exist in syn and anti rotameric forms. We demonstrate that IA, which can exist only in the syn form, is more photostable in alcohols than similar, but non-rigid molecules. This additional photostability enhancement is due to the elimination of a slower channel of excited state deactivation in alcohol complexes, S0 ← S1 internal conversion. The dominant, faster channel of S1 depopulation is the excited state double proton transfer, manifested by the presence of low energy tautomeric fluorescence.


 Please wait while we load your content...
Please wait while we load your content...