An experimental and theoretical study of adenine adsorption on Au(111)†
Abstract
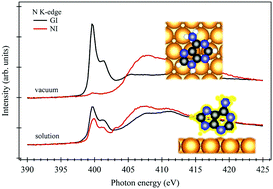
A model study of adenine adsorption on the Au(111) surface is reported for molecular adlayers prepared by evaporation in vacuum and deposition from saturated aqueous solution. The electronic structure and adsorption geometry of the molecular films were studied experimentally by X-ray photoelectron spectroscopy and near edge X-ray absorption fine structure spectroscopy. Adsorption models are proposed for the adlayers arising from the different preparation methods. Density functional theory calculations were used to examine both parallel and upright adenine adsorption geometries, supply additional information on the bond strength, and identify which atom is involved in bonding to Au(111). In the case of deposition in vacuum, the adenine molecule is bound via van der Waals forces to Au(111) with the molecular plane parallel to the surface, consistent with the published scanning tunneling microscopy data on this system. The most stable parallel adenine configuration was found to have an adsorption energy of ca. −1.1 eV using the optB86b-vdW functional. For adenine deposition from aqueous solution, the adlayer is disordered, with molecules in an upright geometry, and with an adsorption energy of ca. −1.0 eV, coordinated via the imino N3 nitrogen atom. The present study contributes to the substantial literature of model studies of adenine on Au(111), complementing the existing knowledge with information on electronic structure, bonding geometry and adsorption energy of this system.
- This article is part of the themed collection: 2018 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...