Molecular rotors confined at an ordered 2D interface†
Abstract
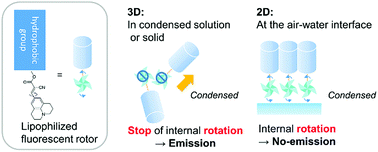
Intramolecular rotation of molecules contained in a two-dimensional monolayer or a three-dimensional collapsed film at an air–water interface was investigated by in situ fluorescence spectroscopy of twisted intramolecular charge transfer (TICT) type 9-(2-carboxy-2-cyanovinyl)julolidine (CCVJ) derivatives. The TICT type molecules, CCVJ-C12 and CCVJ-Chol, that contain a linear alkyl dodecyl chain or a cholesteryl group, respectively, as their hydrophobic group, were designed and synthesized to manipulate them at the air–water interface. These lipophilized molecular rotors showed the general properties of TICT molecules in solutions that the fluorescence intensity increases with increasing viscosity of the solvent, which is induced by inhibition of internal molecular rotations. The molecular rotors CCVJ-C12 and CCVJ-Chol formed monolayers at the air–water interface and in situ fluorescence spectroscopy was performed during the in-plane compression of the monolayers. It was revealed that the monomer emissions were suppressed and only after the collapse of monolayers, excimer emission from both layers consisting of CCVJ-C12 or CCVJ-Chol was observed. Suppressed monomer emission from monolayers suggests that intramolecular rotation is not inhibited in dense ordered monolayers. Furthermore, fluorescence spectroscopy of Langmuir–Blodgett (LB) films indicated that molecular rotations are not inhibited in the monolayer transferred on the solid substrates.
- This article is part of the themed collection: Complex molecular systems: supramolecules, biomolecules and interfaces


 Please wait while we load your content...
Please wait while we load your content...