Efficient extraction of inorganic selenium from water by a Zr metal–organic framework: investigation of volumetric uptake capacity and binding motifs†
Abstract
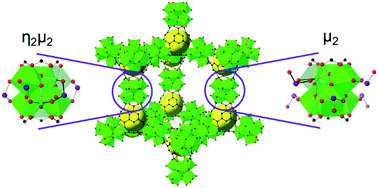
Strict monitoring and control of selenium concentrations in freshwater supplies is critical to safeguarding human health and aquatic life. A handful of previously investigated sorbents exhibit noteworthy gravimetric (mg g−1) Se uptake capacities; however, often display insufficient volumetric (mg cm−3) capacities, thereby requiring large volumes of material for commercial implementation. In pursuit of mitigating this material inefficiency, we investigated the selenite (SeO32−) and selenate (SeO42−) affinity of MOF-808, a Zr-based metal–organic framework with a high density of potential Se oxyanion binding sites. MOF-808 recorded exceptional volumetric and gravimetric Se oxyanion capacities of 133 mg g−1 (127 mg cm−3) and 118 mg g−1 (112 mg cm−3) for aqueous selenite and selenate, respectively. Single-crystal X-ray diffraction studies revealed that selenite and selenate can bind at the MOF node via two distinct binding motifs, an η2μ2 motif in which the oxyanion coordinates to two different metal atoms in a single node, and a μ2 motif in which the oxyanion interacts with only a single metal atom. Furthermore, powder X-ray diffraction (PXRD) patterns and N2 adsorption/desorption isotherms confirm the retention of bulk crystallinity and porosity after the uptake of Se oxyanions.
- This article is part of the themed collection: CrystEngComm 20th volume collection


 Please wait while we load your content...
Please wait while we load your content...