Polar organometallic strategies for regioselective C–H metallation of N-heterocyclic carbenes
Abstract
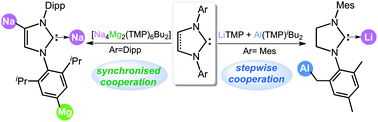
N-Heterocyclic carbenes (NHCs) have become indispensable ligands across a broad swathe of the synthetic and catalytic landscape, not in small part due to their ease of electronic and steric tunability. One of the latest additions to this important family of ligands are anionic NHCs, which have become valuable precursors to access abnormal NHC complexes as well as shown great potential for further NHC functionalisation. Deprotonative metallation has emerged as one of the most versatile methodologies to access anionic NHCs, where judicious choice of reaction conditions and metallating agents can finely tune the regioselectivity of the reaction. This Feature Article focuses on the recent emergence of s-block metal-mediated NHC metallations and the new opportunities this methodology offers.
- This article is part of the themed collection: Editor’s Choice: Main group reagents and catalysts in organic reactions


 Please wait while we load your content...
Please wait while we load your content...