A Cu2Se–Cu2O film electrodeposited on titanium foil as a highly active and stable electrocatalyst for the oxygen evolution reaction†
Abstract
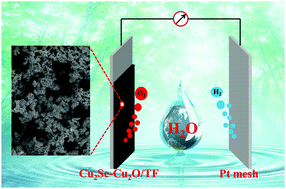
Many nonprecious metal-selenide-based materials have been reported as electrocatalysts with high activity for the oxygen evolution reaction (OER). Herein, a hybrid catalyst film composed of Cu2Se and Cu2O nanoparticles directly grown on Ti foil (Cu2Se–Cu2O/TF) was prepared through a simple and fast cathodic electrodeposition method. Surprisingly, this electrode required a relatively low overpotential of 465 mV to achieve a catalytic current density of 10 mA cm−2 for the OER in 0.2 M carbonate buffer (pH = 11.0). Furthermore, a long-term constant current electrolysis test confirmed the high durability of the Cu2Se–Cu2O/TF anode at a current density of 10 mA cm−2 over 20 h. The XRD, TEM and XPS analysis of the sample after the OER indicated that a CuO protective layer formed on the surface of the Cu2Se–Cu2O catalyst, which effectively suppressed further oxidation of the Cu2Se–Cu2O catalyst during the OER and resulted in sustained catalytic oxidation of water.


 Please wait while we load your content...
Please wait while we load your content...