Abstract
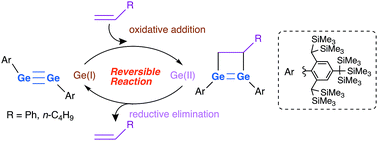
Stable digermynes with sterically demanding Bbt (Bbt = 2,6-[CH(SiMe3)2]2-4-[C(SiMe3)3]-C6H2) or Tbb (Tbb = 4-tBu-2,6-[CH(SiMe3)2]2-C6H2) groups underwent [2+2] cycloadditions with terminal alkenes to give the corresponding 1,2-digermacyclobutenes. In the case of the Bbt-substituted digermyne, the reaction was reversible at room temperature, i.e., the 1,2-digermacyclobutene (Ge(II) species) is susceptible to a facile reductive elimination that affords the corresponding digermyne (Ge(I) species) and the alkene.
- This article is part of the themed collection: Philip Power at 65: an icon of organometallic chemistry


 Please wait while we load your content...
Please wait while we load your content...