Sensitive recognition of ethion in food samples using turn-on fluorescence N and S co-doped graphene quantum dots†
Abstract
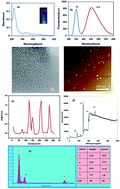
To achieve a fast, sensitive and inexpensive method for the detection of ethion in the presence of Hg2+ ions, a nitrogen and sulfur co-doped graphene quantum dot (N,S/GQD)-based fluorescence sensing system was developed. The fluorescence of N,S/GQDs is sensitively quenched as a result of electrostatic interactions with Hg2+ ions adsorbed onto the negatively charged N,S/GQD surface. Following the introduction of ethion, the fluorescence of the Hg2+-quenched N,S/GQDs is recovered due to the strong coordination between Hg2+ and ethion. Based on the intensities of the recovered fluorescence of the N,S/GQDs, the ethion concentration was easily quantified. The linear response range of this method for ethion was from 19.2 μg L−1 M to 961.2 μg L−1. In this sensing system, not only was the detection limit as low as 8 μg L−1, which was as good as or better than those of similar assays, but the sensing system also displayed good selectivity in detecting ethion in the presence of different physiological molecules and inorganic ions. The proposed sensing method was successfully applied to the detection of ethion in water and food samples and was found to produce encouraging results.


 Please wait while we load your content...
Please wait while we load your content...