Spying on protein interactions in living cells with reconstituted scarlet light†
Abstract
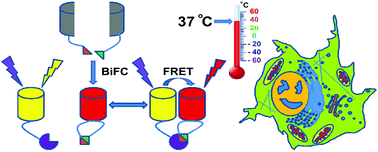
The BiFC (bimolecular fluorescence complementation) assay and BiFC combined with FRET (fluorescence resonance energy transfer) technique have become important tools for molecular interaction studies in live cells. However, the real detection and cellular imaging performances of most existing red fluorescent protein-derived BiFC assays still suffer from relatively low ensemble brightness, high cytotoxicity, the red fluorescent proteins being prone-to-aggregation or severe residual dimerization, inefficient complementation and slow maturation at 37 °C physiological temperature in live mammalian cells. We developed a BiFC assay based on a recently evolved truly monomeric red fluorescent protein (FP) mScarlet-I with excellent cellular performances such as low cytotoxicity, fast and efficient chromophore maturation and the highest in-cell brightness among all previously reported monomeric red fluorescent proteins. In this work, a classic β-Fos/β-Jun constitutive heterodimerization model and a rapamycin-inducible FRB/FKBP interaction system were used to establish and test the performance of the mScarlet-I-based BiFC assay in live mammalian cells. Furthermore, simply by adopting the large-Stokes-shift fluorescent protein mAmetrine as the donor, β-Jun-β-Fos-NFAT1 ternary protein complex formation could be readily and efficiently detected and visualized with minimal spectral cross-talk in live HeLa cells by combining live-cell sensitized-emission FRET measurement with the mScarlet-I-based BiFC assay. The currently established BiFC assay in this work was also shown to be able to detect and visualize various protein–protein interactions (PPIs) at different subcellular compartments with high specificity and sensitivity at 37 °C physiological temperature in live mammalian cells.


 Please wait while we load your content...
Please wait while we load your content...