A combinational molecular design to achieve highly efficient deep-blue electrofluorescence†
Abstract
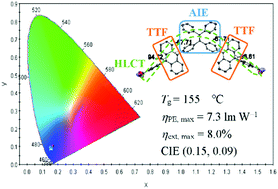
A deep-blue emitter 1-(10-(4-methoxyphenyl)anthracen-9-yl)-4-(10-(4-cyanophenyl)anthracen-9-yl)tetraphenylethene (TPEA) has been successfully prepared by a combinational molecular design, which contains triplet–triplet fusion (TTF) and hybridized local charge transfer (HLCT) characteristics to increase the ratio of triplet excitons used. The tetraphenylethene (TPE) moiety contributes the emitter with an aggregation-induced emission (AIE) property to enhance the solid-state luminescence efficiency. The crystallographic structure shows that the two anthracene groups are twisted from the central TPE moiety, which effectively prevents a bathochromic shift of the emission. In addition, we adopted a donor–acceptor (D–A) structure to improve the charge balance in organic light-emitting diodes (OLEDs). The material possesses high thermal stability with a glass transition temperature (Tg) of 155 °C. Based on all these advantages, a high performance of the non-doped device was achieved with a turn-on voltage (Von) of 2.6 V at a luminance of 1 cd m−2, a maximum power efficiency (ηPE,max) of 11.1 lm W−1, a maximum current efficiency (ηCE,max) of 9.9 cd A−1, and a low current efficiency roll-off even at 1000 cd m−2. Moreover, a deep-blue emission with Commission Internationale de l'Éclairage (CIE) coordinates of (0.15, 0.09), a maximum external quantum efficiency (ηext,max) of 8.0% and the highest ηPE,max of 7.3 lm W−1 among all the TTF and HLCT deep-blue emitters were obtained by doping TPEA into the host of bis-4-[(N-carbazolyl)phenyl]-phenylphosphine oxide (BCPO). These results indicate that the combinational molecular design is promising for highly efficient deep-blue emitters.


 Please wait while we load your content...
Please wait while we load your content...