Defect chemistry and electrical properties of BiFeO3†
Abstract
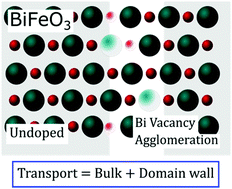
BiFeO3 attracts considerable attention for its rich functional properties, including room temperature coexistence of magnetic order and ferroelectricity and more recently, the discovery of conduction pathways along ferroelectric domain walls. Here, insights into the defect chemistry and electrical properties of BiFeO3 are obtained by in situ measurements of electrical conductivity, σ, and Seebeck coefficient, α, of undoped, cation-stoichiometric BiFeO3 and acceptor-doped Bi1−xCaxFeO3−δ ceramics as a function of temperature and oxygen partial pressure pO2. Bi1−xCaxFeO3−δ exhibits p-type conduction; the dependencies of σ and α on pO2 show that Ca dopants are compensated mainly by oxygen vacancies. By contrast, undoped BiFeO3 shows a simultaneous increase of σ and α with increasing pO2, indicating intrinsic behavior with electrons and holes as the main defect species in almost equal concentrations. The pO2-dependency of σ and α cannot be described by a single point defect model but instead, is quantitatively described by a combination of intrinsic and acceptor-doped characteristics attributable to parallel conduction pathways through undoped grains and defect-containing domain walls; both contribute to the total charge transport in BiFeO3. Based on this model, we discuss the charge transport mechanism and carrier mobilities of BiFeO3 and show that several previous experimental findings can readily be explained within the proposed model.


 Please wait while we load your content...
Please wait while we load your content...