Photoswitchable polyfluorophores based on perylenemonoimide–dithienylethene conjugates as super-resolution MitoTrackers†
Abstract
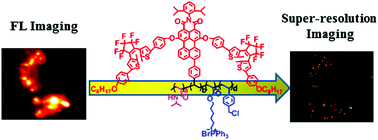
We design and synthesize a novel type of photoswitchable polyfluorophores based on perylenemonoimide–dithienylethene (PMI–DTE) conjugates, which are modified by triphenylphosphonium and benzyl chloride for mitochondria-targeting super-resolution imaging. The strong emitter is the PMI unit plus two DTE units as the photoswitching quencher, while triphenylphosphonium and vinylbenzyl chloride act as twofold mitochondria-targeting units and the incorporation of N-isopropylacrylamide (NIPAM) affords the hydrophilicity of polymeric fluorophores. The super-resolution mitochondria-targeting imaging agents are created with strong photoswitchable fluorescence in aqueous solution because the emitter units are isolated from each other by the surrounding NIPAM units throughout the polymer chains. The as-synthesized MitoTrackers show strong fluorescence and dramatic fluorescence-switching behavior with a fluorescence switching ON/OFF ratio of 30 : 1 in aqueous solution. Fluorescent switching enables mitochondria-targeting super-resolution imaging of polyfluorophores with sub-30 nm spatial resolution in imaging subcellular organelles. The conceptual design of photoswitchable polyfluorophores presented here is promising to guide the development of future-generation super-resolution bioimaging agents.


 Please wait while we load your content...
Please wait while we load your content...