Influence of ligand shell ordering on dimensional confinement of cesium lead bromide (CsPbBr3) perovskite nanoplatelets†
Abstract
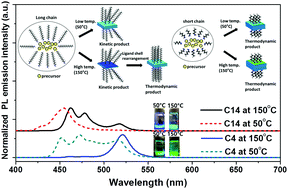
The perovskite cesium lead bromide (CsPbBr3) has emerged as an attractive thermally and chemically robust alternative to hybrid lead perovskite halides and analogues of this material show excellent tunability of exciton binding energies, high absorption cross-sections, and intense photoluminescence. Dimensional reduction, particularly in proximity of the Böhr exciton radius, allows for substantial tunability of the photophysical properties of this material as a result of quantum confinement. The use of surface passivating ligands, particularly alkylammonium cations, has been developed as a means of inducing directional growth and facilitates dimensional confinement of the obtained perovskite nanocrystals. Here, we demonstrate that the crystalline order of the ligand-shell assembly, as dictated by the length of the alkyl chains, the degree of branching, the reaction temperature, and ligand concentration, strongly influences the extent of dimensional confinement attainable for the perovskite nanoplatelets. The spatial extent of the ligand shell and the degree of ordering of ligand molecules greatly impact the diffusion and addition of monomeric species. The interplay between enthalpic stabilization from crystalline packing and entropic loss from loss of configurational degrees of freedom provides substantial opportunity to tune the parameter space as a function of ligand structure and reaction variables. Mechanistic understanding of thermodynamic and kinetic regimes provides a means to rationally optimize synthetic parameters to obtained desired dimensionality and thus allows for control over nanocrystal thickness in precise increments.


 Please wait while we load your content...
Please wait while we load your content...