Thermodynamic considerations on interfacial reactivity concerning carrier transport characteristics in metal/p-Zn3P2 junctions
Abstract
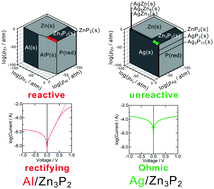
Carrier transport across a metal/semiconductor interface plays a key role in electronic devices. It is generally affected by some interactions at interfaces, such as atomic interdiffusion and formation of intermediate compounds; however, it is hard to consider all possible chemical reactions at the interface. In this work, we adopted a chemical potential diagram for comprehensive discussions on chemical reactions at the interfaces between the metal (M) and p-type zinc phosphide (p-Zn3P2), which is a promising candidate as an absorber in thin-film solar cells. We revealed that Schottky or Ohmic behavior depended on the reaction at the interfaces through the investigations in Al/p-Zn3P2 and Ag/p-Zn3P2 junctions. The interdiffusion was observed at the interface of Al/p-Zn3P2, while the interfacial structure in Ag/p-Zn3P2 was not changed by annealing. Such behaviors were thermodynamically expected from the chemical potential diagrams and determined the transport characteristics considering the similar work functions of Al and Ag. Furthermore, the discussions on other M–P–Zn (M = Au, In, Mn, Cu) systems based on chemical potential diagrams clarified that phosphide semiconductors as intermediate compounds were formed in the M/p-Zn3P2 junctions, which show Schottky behavior, and that Ohmic M/p-Zn3P2 junctions are stable. Therefore, it is concluded that we should discuss the band structures in heterojunctions under the considerations on reactivity at the interfaces, which is comprehensively understood using chemical potential diagrams.


 Please wait while we load your content...
Please wait while we load your content...