Self-assembly of polyoxometalate/reduced graphene oxide composites induced by ionic liquids as a high-rate cathode for batteries: “killing two birds with one stone”†
Abstract
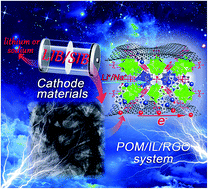
The relatively low capacity and poor rate performance of cathodes restrict the development of rechargeable batteries and must be settled urgently due to the ever-growing need of energy storage. Here, we report a new cathode system that produces polyoxometalates (POMs)/ionic liquid (IL)/reduced graphene oxide (RGO) composites, denoted as PIG, by a self-assembly method in which IL plays the role of “killing two birds with one stone”. IL not only facilitates the formation of heterogeneous nanocrystalline composites but also acts as the template reagent to feature the morphology of homogeneous nanobelts on the RGO. The PIG system provides us with a theoretical model at the molecular level to give a detailed comparison of a three-dimensional open skeleton formed by different transition metal linkers and a vanadium cage, on the performance of the cathode in batteries. Finally, the targeted composite Mn3V19-HIL/RGO-1 shows good cycling stability and the best ultrafast rate capabilities (121 mA h g−1 at 5000 mA g−1 and 73 mA h g−1 at 2000 mA g−1 for lithium and sodium ion batteries) of POMs-based composites. Furthermore, the PIG system provides a platform for the design of POMs, and even anionic clusters-based composites in the energy conversion and storage, thus giving access to their versatile architectural design and applications.


 Please wait while we load your content...
Please wait while we load your content...