Platinum group metal-free NiMo hydrogen oxidation catalysts: high performance and durability in alkaline exchange membrane fuel cells†
Abstract
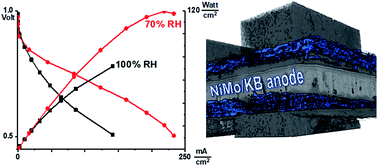
We introduce a new platinum group metal-free (PGM-free) hydrogen oxidation electrocatalyst with superior performance in anodes of alkaline exchange membrane fuel cells (AEMFCs). A carbon-supported bimetallic nickel–molybdenum catalyst was synthesized by thermal reduction of transition metal precursors on the surface of a carbon support (KetjenBlack 600J). The mass-weighted activity of 4.5 A gMe−1 determined in a liquid electrolyte 0.1 M NaOH using a rotating disk electrode (RDE) technique is comparable to the value reported for Pd/C with a comparable particle size under similar conditions. This NiMo/KB catalyst was integrated in a membrane electrode assembly (MEA) using an alkaline exchange membrane and ionomer. Single AEMFC tests performed in a H2/O2 configuration resulted in a record power density output of 120 mW cm−2 at 0.5 V, the MEA was found to be durable under the conditions of potential hold of 0.7 V for 115 h. For the first time, operando X-ray computed tomography (CT) experiments were performed demonstrating liquid water formation at the PGM-free anode during cell operation, and in situ ambient pressure X-ray photoelectron spectroscopy (APXPS) and X-ray absorption spectroscopy (APXAS) were used to study the role of molybdenum in hydrogen adsorption.


 Please wait while we load your content...
Please wait while we load your content...