Graphitic carbon nanofiber growth from catalytic-metal organic frameworks & their electrochemical double layer properties†
Abstract
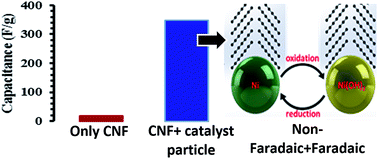
Metal–organic frameworks have been widely studied as a template to nanoporous or well-structured carbon materials; however, utilizing the catalytic properties of MOFs to grow graphitic one dimensional nanostructures has been rarely reported. We have studied the evolution of carbon structures produced by transition metal doping (Ni in our case) in a Zn-based MOF and subjecting the MOF to a typical chemical vapour deposition process. Structures ranging from fine to thick graphitized carbon nanofibers are produced systematically at a relatively low temperature of 650 °C by increasing the Ni concentration in the MOF. A general trend of an increase in the nanofiber diameter with an increase in the Ni concentration and the formation of plate-like graphitic structures suggest that the catalyst particles undergo coalescence and impact the morphology of carbon structures produced. Without any further modification, the carbon nanofibers show modest electrical double layer (18 F g−1) behaviour in 1 M Na2SO4, but quite interestingly, the finely divided Ni particles undergo faradaic redox reactions in 1 M KOH and boost the capacitance to 348 F g−1. Our work suggests that our approach of utilizing catalytic MOFs for the growth of well-defined carbon nanostructures can serve more than one purpose towards the synthesis of hybrid materials for energy storage.


 Please wait while we load your content...
Please wait while we load your content...