A nickel coordination supramolecular network synergized with nitrogen-doped graphene as an advanced cathode to significantly boost the rate capability and durability of supercapacitors†
Abstract
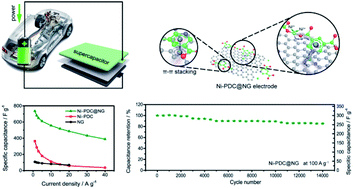
The strategy of developing new electrode materials, which integrate well with carbon materials or conducting polymers to form a robust synergistic composite, can distinctly boost the electrochemical performance of supercapacitors (SCs). In this study, a novel Ni coordination supramolecular network (CSN) of [Ni2(3,5-PDC)2·(H2O)8·(H2O)2]n (denoted as Ni-PDC, 3,5-PDC = 3,5-pyridinedicarboxylic acid) was successfully integrated with nitrogen-doped graphene (NG) via a one-step hydrothermal method, forming a superior composite electrode material of Ni-PDC@NG. The Ni-PDC@NG electrode showed a high specific capacitance of 735 F g−1 at a current density of 1 A g−1 and an outstanding cycling stability (85.3% capacitance retention after 14 000 cycles); furthermore, it also maintained a good rate capability by exhibiting a capacitance retention of 53% even at a high current density of 40 A g−1. Significantly, a SC device was assembled with the Ni-PDC@NG electrode as the cathode and an activated carbon electrode as the anode, which not only exhibited a high energy density of 21.7 W h kg−1 at a power density of 801 W kg−1, but also retained 45.2% of its initial energy density even at a high power density of 16 036 W kg−1. Besides, this as-fabricated device exhibited excellent long-term durability with 97.9% capacitance retention after 6000 cycles.


 Please wait while we load your content...
Please wait while we load your content...