Efficient removal of Pb(ii) from water using magnetic Fe3S4/reduced graphene oxide composites†
Abstract
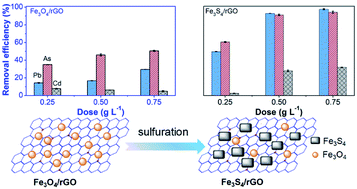
Nanostructured metal sulfides hold great promise for adsorption and catalysis applications, but it remains challenging for them to achieve highly efficient decontamination and facile separation of heavy metal ions from water. Herein, Fe3S4/reduced graphene oxide composites (Fe3S4/rGO) were successfully prepared and utilized for the removal of Pb(II) from water. Uniform Fe3O4 nanoparticles were firstly dispersed on the rGO, and employed as sacrificial materials for a facile sulfuration approach. This gave rise to the fabrication of Fe3S4 nanoparticles (Fe3S4 NPs) with an average size of 14.3 nm. The resultant Fe3S4/rGO presented an evidently greater adsorption capacity (285.71 mg g−1) and advantages in driving fast adsorption of Pb(II) in comparison with Fe3O4/rGO (106.27 mg g−1). A remarkable selectivity for Pb(II) was achieved in the presence of coexisting cations and anions, and the addition of humic acid promoted the removal of Pb(II) by Fe3S4/rGO. The enhanced performance of Fe3S4/rGO was mostly facilitated by synergetic contributions of the strong and selective Pb–S interactions between Pb(II) and small Fe3S4 NPs associated with the surface adsorption. Remarkably, a single treatment of smelting wastewater with Fe3S4/rGO effectively reduced Pb(II) concentration below the drinking water standard that is recommended by the U.S. Environmental Protection Agency, along with a surprisingly high removal efficiency toward arsenic (96.18%). However, Fe3O4/rGO merely exhibited a low removal rate of 29.59% for Pb(II). The efficient removal performance of Fe3S4/rGO exhibited its great potential in the remediation of heavy metal polluted water via simultaneously taking advantage of magnetic properties and a high affinity for heavy metals.


 Please wait while we load your content...
Please wait while we load your content...